Abstract
Background: Since the definition of MDS their classification relied on cytomorphology determining dysplasia, cytopenia and blast count. The revised 4th edition of the WHO classification encompasses only one genetically defined entity (MDS with isolated del(5q)). In the 5th edition of the WHO classification 2 additional entities are defined by genetics: MDS with low blasts and SF3B1 mutation and MDS with biallelic TP53 inactivation. All other are classified still based on morphology into i) hypoplastic MDS, ii) MDS with low blasts and iii) MDS with increased blasts.
Aim: As blast counting is subjective and the boundary between low and elevated blasts is arbitrary, we set out to propose a classification for MDS based solely on genetic abnormalities obviating the need for blast counting.
Patients and Methods: The cohort comprised 735 de novo MDS. The diagnosis was established following WHO classifications. In all cases chromosome banding analysis and WGS (100x, 2x151bp, NovaSeq) were performed. Variants were called with Strelka2, Manta and GATK using a tumor w/o normal pipeline. For all cases overall survival data was available (median follow-up: 9.3 years).
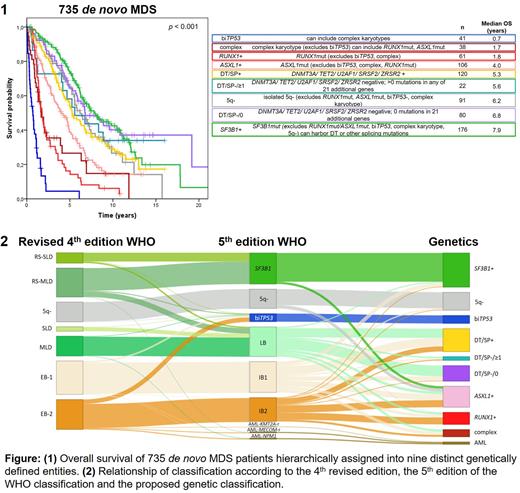
Results: We categorized our cohort 1) according to WHO revised 4th edition, 2) 5th edition, 3) according to blast count (<5%, 5 to <10%, 10 to <20%), and 4) according to numbers of mutations. For all 4 categorizations prognostic differences between subgroups was confirmed. Next, we performed cluster analyses to identify genetic abnormalities associated with blast count. While SF3B1 mutations and isolated del(5q) were associated with low blast count, mutations in ASXL1 and RUNX1 were associated with higher blast count (for all p<0.0001). Further, we evaluated the genomic landscape for associations of genetic parameters. RUNX1 mutations and ASXL1 mutations were highly associated with spliceosome mutations. 49/62 (79%) RUNX1mut MDS and 97/153 (63%) ASXL1mut MDS harbored also spliceosome mutations. On the other hand RUNX1mut, ASXL1mut and spliceosome mutations rarely co-occurred with biallelic TP53 inactivation or complex karyotype (only 5/41 cases and 19/78 cases). 102/735 (14%) cases harbored neither a del(5q), a complex karyotype, biallelic TP53 inactivation nor a mutation in SF3B1, SRSF2, U2AF1, ZRSR2, DNMT3A, TET2, RUNX1 or ASXL1. In 20 of these at least one mutation was found in 21 additional genes known to be mutated in MDS. Based on these data 9 genetically defined non-overlapping hierarchical subgroups (definitions of the respective prior subgroups are exclusion criteria for the following ones) are proposed: 1. biallelic TP53 inactivation 2. complex karyotype, 3. RUNX1 mutated, 4. ASXL1 mutated, 5. del(5q), 6. SF3B1 mutated, 7. mutated DNMT3A, TET2, U2AF1, SRSF2, and/or ZRSR2, 8. presence of at least one mutation in one of 21 other genes recurrently mutated in MDS, 9. none of the genetic markers present that define any of the previous entities. Survival analyses revealed marked differences between these biologically defined entities (figure 1). In univariate cox regression analyses including all 9 genetic categories and blast count the categories biallelic TP53 inactivation, complex karyotype, RUNX1mut, ASXL1mut, SF3B1mut, none of the genetic markers and blast count <5% vs ≥5% and <10% vs ≥10% were significantly associated with OS (for all p<0.002). In multivariate analyses both blast count cut offs did not show a significant impact, while the categories biallelic TP53 inactivation, complex karyotype, RUNX1mut, ASXL1mut, SF3B1mut were independently associated with OS (for all p<0.05). In figure 2 the relationship of classification according to the 4th revised, the 5th edition of the WHO and the purely genetically based classification as proposed here is depicted.
Conclusions: Based on karyotype and mutation status of 9 genes complemented by 21 genes in 10-15% of cases, MDS can be separated into 9 biologically distinct subgroups, reflecting biology better than blast counts. These are associated with significant differences in overall survival. RUNX1 and ASXL1 define distinct subgroups within spliceosome mutated MDS potentially driving progression. Given the poor survival of the MDS subgroups with biallelic TP53 inactivation, complex karyotype and RUNX1 mutation it has to be further evaluated whether or not these MDS subtypes should be combined with AML harboring the respective genetic abnormalities for treatment approaches.
Disclosures
Haferlach:MLL Munich Leukemia Laboratory: Current Employment, Other: Ownership. Huber:MLL Munich Leukemia Laboratory: Current Employment. Mueller:MLL Munich Leukemia Laboratory: Current Employment. Baer:MLL Munich Leukemia Laboratory: Current Employment. Hoermann:MLL Munich Leukemia Laboratory: Current Employment. Meggendorfer:MLL Munich Leukemia Laboratory: Current Employment. Hutter:MLL Munich Leukemia Laboratory: Current Employment. Kern:MLL Munich Leukemia Laboratory: Current Employment, Other: Ownership. Haferlach:Munich Leukemia Laboratory: Current Employment, Other: Part ownership.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal